Wet strength agents (WSA) are important functional additives in papermaking. Since neutral manufacturing became a standard in industry, Polyamidoamineepichlorohydrine (PAE) resins have been the most relevant class of wet strength agents. Consumer tissue products represent the biggest group of wet strengthened papers today.
During the synthesis of the PAE resin there is a potential for the formation of hazardous by-products such as 1,3-Dichloro-2-propanol (DCP) and 3-Monochloropropane-1,2-diol (MCPD) deriving from hydrolysis of the Epichlorohydrine (ECH) reactant.
The article gives an overview of the current regulatory situation and how the tightened requirements of the revised EU Ecolabel for paper products which becomes effective as of January 1st, 2019 will affect the properties of wet strength agents in tissue manufacturing.
Formation of by-products in the wet strength resin and their control
The monomeric by-products 1,3-Dichloro-2-propanol (DCP) and 3-Monochloropropane-1,2-diol (MCPD) are formed during resin-synthesis [1]:
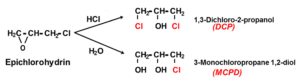
In the early 2000´s, both DCP and MCPD have been classified as carcinogen, MCPD as genotoxic moreover, which is why miscellaneous limits for the DCP and MCPD content in food contact paper and board as well as the wet strength agents have been set. The chemical industry took intense R&D efforts for significant reduction of DCP & MCPD contents in the PAE-resins during the past decades. Figure 2 shows the characteristics of the different wet strength agent generations:
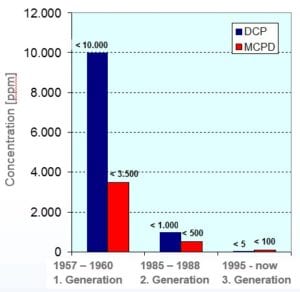
The formation of DCP and MCPD cannot be avoided during the resin synthesis, but their content can be adjusted to the required level by the following methods:
- A very cost-effective way of reducing these by-products is an in process alkaline treatment. This method enables producers to reduce the DCP/MCPD content of a wet strength agent with 20 % active matter to a total level of below 500 ppm. Alkaline treated products are sometimes called “G2.5” wet strength agents.
- Genuine G3 (generation 3) WSAs are commonly post-treated via ion exchange or membrane filtration. It is possible to eliminate the DCP/MCPD content in the wet strength agent to a total level of significantly below 25 ppm with these technologies. A further asset of the ion exchange treatment is the significant reduction of AOX and excessive chloride in the wet strength agents, which supports the reduction of AOX in mills´ effluents.
REGULATORY FRAMEWORK
In the light of rising concerns about a potential migration of DCP/MCPD into the food chain deriving from food contact paper and board, in 2001, the German Federal Institute of Risk Assessment, BfR, has implemented limits for DCP and MCPD that may be extracted with water from a paper sample in their recommendations XXXVI. for food contact paper and board grades:
“1,3-Dichloro-2-propanol must not be detectable in water extract of the finished product (detection limit 2 μg/l). The transfer of 3-monochloro-1,2-propanediol into the water extract of the finished products must be as low as technically achievable, a limit of 12 μg/l must not be exceeded in any case.”
Table 1 gives an overview of the different guidelines for sampling and preparation of the aqueous extract in misc. paper grades:
| Paper grade | Sample size (g/l) | Extraction method | Water temperature | 1,3-DCP in aqueous extract (µg/l) | 3-MCPD in aqueous extract (µg/l) |
| Teabags | 40 | EN 647 | Hot | <2 | 12 |
| Coffee filter | 40 | EN 647 | Hot | <2 | 12 |
| Food packaging papers | 40 | EN 645 | Cold | <2 | 12 |
| Kitchen towel | 4 | EN 645 | Cold | <2 | 12 |
Table 1: Extraction methods for the determination of 1,3-DCP and 3-MCPD in paper samples
The sampling and extraction methods reflect the real exposure. Hence, a significantly higher DCP and MCPD content in kitchen towel grades is tolerable, which is why consumer tissue grades in Europe are mainly manufactured with G 2 and G2.5 wet strength agents, whereas food packaging and filter grades require the particular use of G 3 products.
The recommendations XXXVI. of German BfR [2] have been adopted on European level, e.g. in the 2012 version of the CEPI “Industry Guideline for the compliance of Paper & Board Materials and Articles for food contact” [3] or the 2004 “Policy statement concerning tissue paper, kitchen towel and napkins” [4], issued by the EC Committee of Experts on materials coming into contact with food.
Meeting the requirements of the BfR XXXVI. recommendations is therefore an important prerequisite for producers to comply with the regulatory framework of the European market.
NON-GOVERNMENTAL GUIDELINES
Relevant Ecolabel systems in Europe contain direct limits for DCP/MCPD in the wet strength agents. The limits set for DCP/MCPD in the “EU Ecolabel for Tissue Paper (2009/568/EC)” and the ”Nordic Ecolabelling of Tissue Paper (Nordic Swan)” say, that “Wet strength agents must not contain more than 0.7 % of the chloro-organic-substances ECH, DCP and MCPD, calculated as the sum of the three components and related to the dry content of the wet strength agent”.
In practice the limit can be particularly associated with DCP/MCPD as up-to-date WSAs do not contain ECH after production.
According to the technical report 3.0 of the EC´s Joint Research Centre, a revision of the EU-Ecolabel criteria for Paper products is in preparation [5]. The current criteria document 2009/568/EC will be repealed by December 31st, 2018.
The revision is more restrictive regarding the maximum content of ECH, DCP and MCPD, as the combined content must not exceed 0.35 % of the active solids in future. Table 2 gives an overview of the max. combined content of ECH/DCP/MCPD in wet strength agents with different active solids contents for Tissue papers:
Dry content WSA(%) |
Nordic Ecolabela
(ppm) |
EU-Ecolabelb
(ppm) |
Revised 2019
EU-Ecolabel (ppm) |
| 12.5 | 875 | 875 | 437.5 |
| 15 | 1,050 | 1,050 | 525 |
| 20 | 1,400 | 1,400 | 700 |
| 22 | 1,540 | 1,540 | 770 |
| 25 | 1,750 | 1,750 | 875 |
a) Nordic Ecolabelling of Tissue Paper, Version 5.6
b) 2009/568/EC
Table 2: Maximum tolerable combined contents of ECH, 1,3-DCP and 3-MCPD in wet strength agents
Tissue manufacturers who need to comply with the EU-Ecolabel will have to reconsider their wet strength programs after the tighter requirements of the revision became effective.
MILL CASES
Study 1 – Excessive MCPD contents in kitchen towel after increase of short fiber ratio
Background: A manufacturer of virgin fiber based consumer tissue products for private label couldn´t meet the BfR requirements at higher wet strength agent dosages after increase of the short fiber ratio from 40% to 70 % resulting in an increase of WSA dosage by 30 %. A System overview is shown in Table 3.
| Machinery: | One layer Crescent former machine, working width 2.76 m |
| Operational speed: | 1,300 – 1,650 m/min |
| Grades: | Kitchen towels, toilet paper, hankies |
| Furnish: | 30 – 40 % NBSK, 60 – 70 % BEKP |
| Wet-strength-chemistry: | G2 PAE resin, 20 % dry solids, ca. 1,100 ppm DCP/MCPD, dosage 15 – 35 kg/t |
Table 3: System overview of mill study 1
Objective: Restoring compliance with BfR requirements.
Approach: A G2.5 WSA with reduced contents of chloro-organic compounds
(Giluton 20 XP, 20 % dry solids, ca. 650 ppm DCP/MCPD).
Achievements: Giluton 20 XP was found suitable to safely control DCP/MCPD in the aqueous paper extracts below the limits. Furthermore, Giluton 20 XP has shown similar performance compared to the G2 WSA.
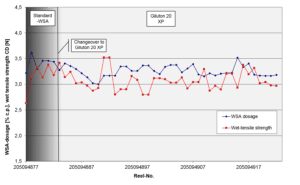
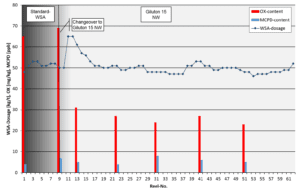
Study 2 – High OX-contents in TCF-grades
Background: A manufacturer of virgin fiber based premium tissue products couldn´t meet the market requirement of max. 30 mg/kg OX (organically bound chlorine) in TCF-grades. A system overview is shown in Table 4.
| Machinery: | Three layer C-wrap former, TAD dryer, working width 5.30 m |
| Operational speed: | 1,200 – 1,350 m/min |
| Grades: | toilet paper, kitchen towel, hankies |
| Furnish: | 60 – 70 % NBSK, 30 – 40 % BEKP |
| Wet-strength-chemistry: | G2.5 PAE resin, 20 % dry solids, ca. 2,500 ppm AOX, ca. 260 ppm DCP/MCPD, dosage 40 – 60 kg/t |
Table 4: System overview of mill study 2
Objective: Meeting the target of < 30 mg/kg OX in finished paper.
Approach: A high-performance G2.5 WSA with sign. reduced AOX contents and low DCP/MCPD content (Giluton 15 NW, 15 % dry solids, ca. 250 ppm DCP/MCPD, AOX ca. 650 ppm).
Achievements: OX in paper has been on target after implementation of Giluton 15 NW. The dosage remained on a similar level leading to 15 % cost savings.
CONCLUSION
Thanks to intense R&D efforts and their resulting improvements regarding efficiency and sustainability, PAE resins represent the best in class technology for the manufacturing of wet-strengthened papers such as tissue. A broad range of available products with adapted purity levels enables papermakers to meet the requirements of the current regulatory framework as well as the latest requirements of different Ecolabel systems on the lowest possible cost-in-use level.
REFERENCES
- Kramer, Gunnar, Stumm, Dominik, chapter 4.6.4, Wet Strength resins, in: Holik, Herbert (ed.), Handbook of Paper and Board, Volume 2, Wiley-VCH, Weinheim (2013)
- bfr.bund.de, Database BfR Recommendations on Food contact materials, Recommendation XXXVI.0 Paper and Board for Food contact, Berlin (2017)
- CEPI-Industry Guideline for the Compliance of Paper & Board Materials and Food contact articles, page 2, Confederation of European Paper Industries, Brussels (2012)
- Public Health Committee, chapter 1.2.2.6, Wet Strengths, in: Policy Statement concerning Tissue Paper, Kitchen Towels and Napkins, Council of Europe (2004)
- Malgorzata Kowalska et al., chapter 5.4.1.1, Criteria proposal, in: Revision of the EU Ecolabel criteria for Copying and Graphic Paper and Newsprint Paper, EC (2018)
Kurita is your competent partner for comprehensive solutions. Our technical sales representatives and application technologists provide you locally with reliable customised services and solutions for your operations.
We take the continuous improvement of your process to heart. Our aim is to support you in reducing water and chemicals consumption and adding value to paper while keeping environment and sustainability in the focus. For good reasons, Kurita is one of only 30 enterprises which are listed in the „NAI“ (Nature-Stock-Index).
Visit our Tissue and Towel page
Contact us in case you need support or wish more information. Our qualified experts will be glad to advise you.